Is Lemon Juice a Good Conductor of Electricity
If the bulb is lit then it indicates that the solution is a conductor of electricity. Lemon juice contains citric acid.
Only some liquids can conduct electricity.
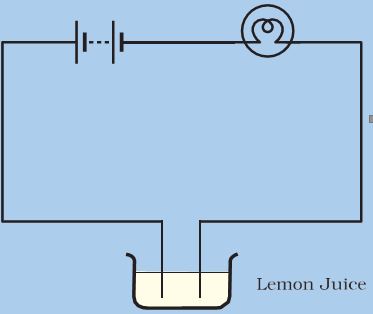
. In this experiment we will see that the bulb will lighted up. Lemon is basically a citrus fruit which contains an appreciable quantity of citric acid. The citric acid in the lemon will be acting as an electrolyte a solution that can conduct electricity.
What is an LED. The flow of ions or charges from one terminal of electrode to another causes electricity to be conducted through the liquid. Vinegar is a sour liquid.
Vinegar is a sour liquid. Acids give H ions when dissolved in water these free cations and anions make lemon juice a good conductor. When the ends of a tester is dipped in lemon juice or vinegar the bulb glows.
Lemon is a citrus fruit containing citric acid. This process indicates that lemon juice and vinegar both are good conductors of electricity. Lemon juice is a good conductor of electricity.
When lemon juice is added to water the acid dissociates into anions and cations which are charged. The acid in the lemon is dissolved into positive and negative ions in its natural water. This acid furnishes H ions and we know that motion of ions charged species causes the flow of current.
Hence they can conduct electricity as these charged particles are able to flow inside the acid. Since it releases H and CH3COO- ions movement of these ions in the solution aids in the conduction of electricity. Now heat the solution and turn on the power supply.
Vinegar is mainly used as a preservative and a flavouring agent. Lemon juice contains citric acid which is a strong electrolyte. Perform the same activity with vinegar you will get the same result.
An activity can be done to show that lemon juice and vinegar are good conductor of electricity. Show with the help of a diagram that lemon juice and vinegar are good conductors of electricity. According to the California Science Center lemon juice can both conduct and produce electricity.
Even though lemon as well as vinegar are weak conductors of electricity. The liquids can be tested for conductor of electricity. The free ends of a tester are dipped in liquid to be tested and then bulb is observed if it glows it shows that the liquid is good conductor otherwise it is a poor conductor.
How would you classify lemon juice- a good conductor or a poor conductor of electricity. Hence we can say that vinegar is a good conductor of electricity. Why is it preferred to other type of bulbs.
Thus lemon juice is a good conductor of electricity due to. Because acids break up into charged anions and cations when dissolved in water they conduct electricity because the charged particles are able to flow within the acid. When the vinegar dissociates in water the following reaction occurs as shown below.
Lemon juice conducts electricity but distilled water does not. The electrolytes in lemon produce electricity by allowing two metals to react with each other. So we can say that lemon juice is the conductor of electricity.
Lemon juice is a good conductor of electricity because it contains citrus acid having H ions which are responsible for the conduction.
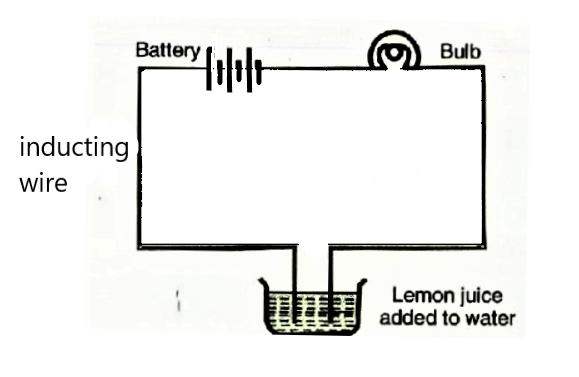
What Happens When We Add A Pinch Salt Or Some Lemon Juice In Distilled Water To Test Its Conduction Of Electric Current Snapsolve
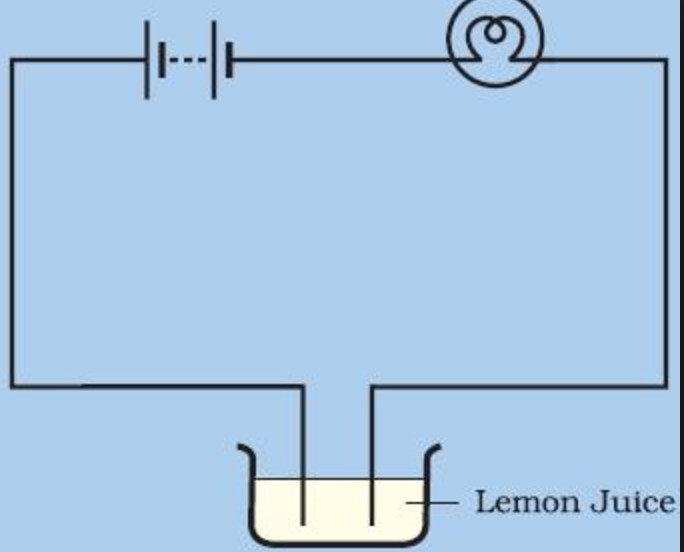
Weak Conductors Of Electricity Chemical Effect Of Electric Current Class 8

Show That Lemon Juice And Vinegar Are Good Conductors Of Electricity Snapsolve
Comments
Post a Comment